Halide precursors
Hafnium Tetrachloride HfCl4
-
Melting point 432°C (vapor pressure 44.4 atm)
-
Sublimation temperature 315°C (1 atm)
-
Condensed phase density 3.86 g/cm3 (at 25°C)
-
Molar mass 320.3 g/mol
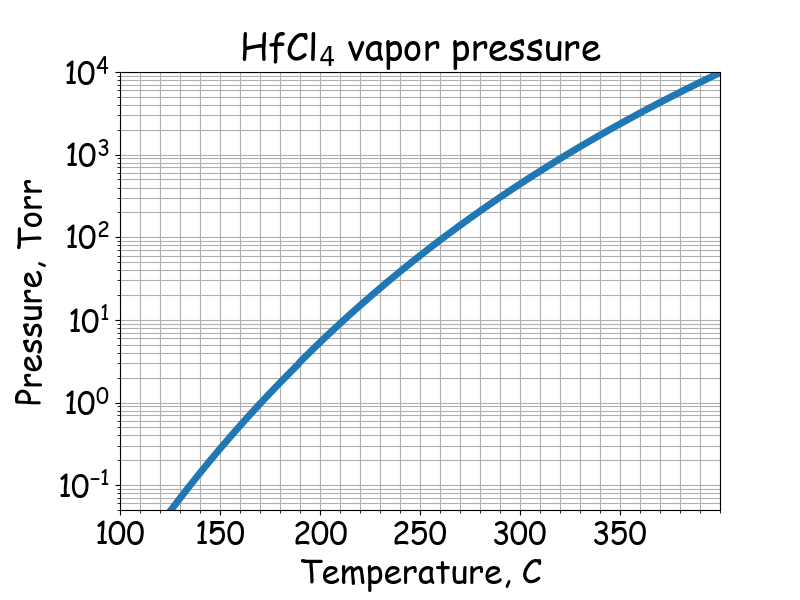
Saturated vapor pressure.
# T -- temperature, C
# P -- saturated vapor pressure, Pa
# Thermochimica acta 244 (1994): 249-256.
if 125 <= T <= 227:
P = 10 ** (13.64 - 5112/ (T + 273.15))
# P -- saturated vapor pressure
# The Journal of Physical Chemistry, 62(3), 319-322.
if 190 <= T <= 400:
P = 10**(11.712 - 5197.0/(T + 273.15))
Hafnium Tetraiodide HfI4
-
CAS Registry Number: 13777-23-6
-
Molar mass: 686.11 g/mol
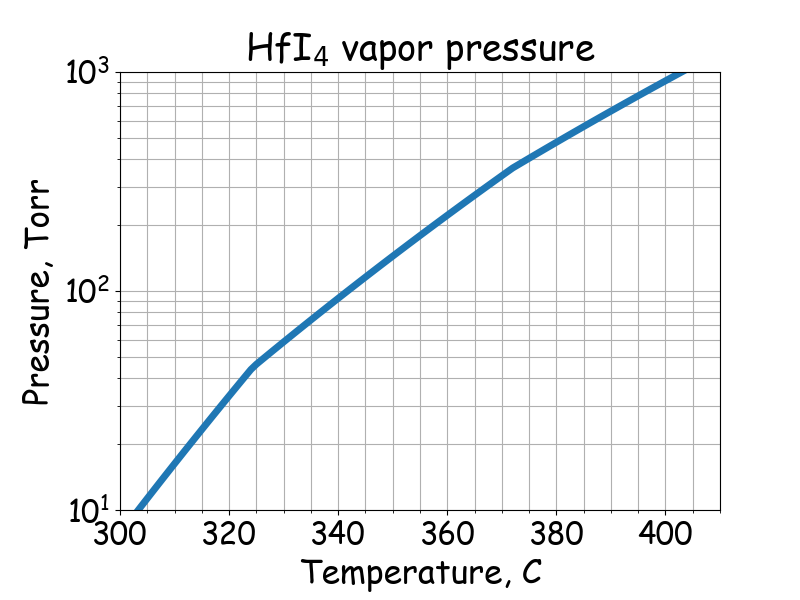
Saturated vapor pressure.
# T -- temperature, C
# P -- saturated vapor pressure, Torr
# Stevenson, F. D., Wicks, C. E., & Block, F. E. (1963)
# gamma-solid
if 372 <= T <= 405:
P = 10 ** (12.13 - 6173/(T + 273.15))
# beta-solid
if 325 <= T <= 372:
P = 10 ** (13.97 - 7360/(T + 273.15))
# alpha-solid
if 302 <= T <= 324:
P = 10 ** (19.56 - 10700/(T + 273.15))
Bibliography
-
Stevenson, F. D., Wicks, C. E., & Block, F. E. (1963). Vapor pressure of tungsten (VI) chloride and hafnium (IV) iodide by a metal diaphragm technique (No. BM-RI-6367). Bureau of Mines, Albany, OR (USA). Albany Metallurgy Research Center.
-
Tangri, R. P., and D. K. Bose. "Vapour pressure measurement of zirconium chloride and hafnium chloride by the transpiration technique." Thermochimica acta 244 (1994): 249-256.
-
Palko, A. A., Ryon, A. D., & Kuhn, D. W. (1958). The vapor pressures of zirconium tetrachloride and hafnium tetrachloride. The Journal of Physical Chemistry, 62(3), 319-322.