Halide precursors
Niobium Fluoride NbF5
-
Melting point: 78.9°C
-
Boiling point: 233.3°C
-
Condensed phase density: 3.29g/cm3 (at 25°C), 2.69g/cm3 (at melting point)
-
Molar mass: 187.9g/mol
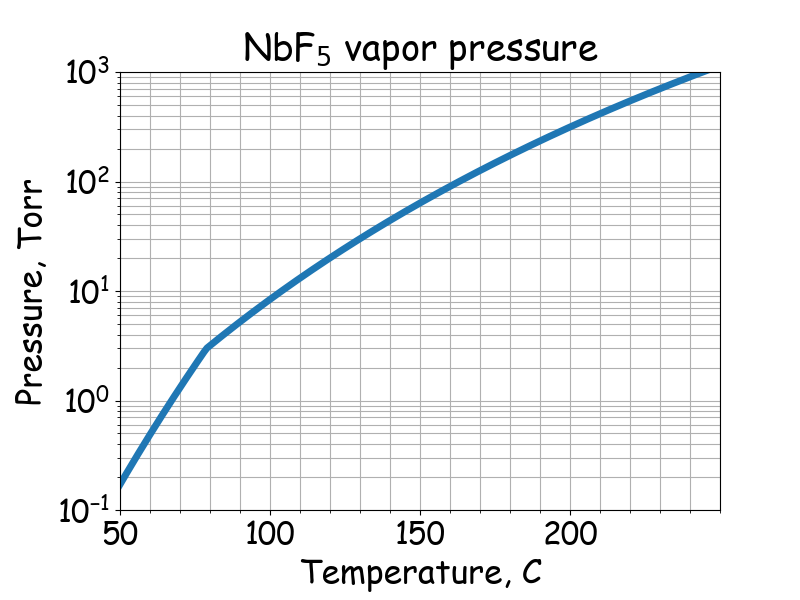
Saturated vapor pressure.
# T -- temperature, C
# P -- saturated vapor pressure, Torr
# Journal of the American Chemical Society, 74(14), 3464-3466.
if 78.9 <= T <= 255:
P = 10 ** (8.3716 - 2779.3 / (T + 273.15))
if 20 <= T <= 78.9:
P = 10 ** (14.397 - 4900 / (T + 273.15))
Niobium Chloride NbCl5
-
Melting point 205°C
-
Boiling point 247.5°C
-
Condensed phase density 2.75 g/cm3 (at 25°C), 2.07 g/cm3 (at melting point)
-
Molar mass 270.2 g/mol
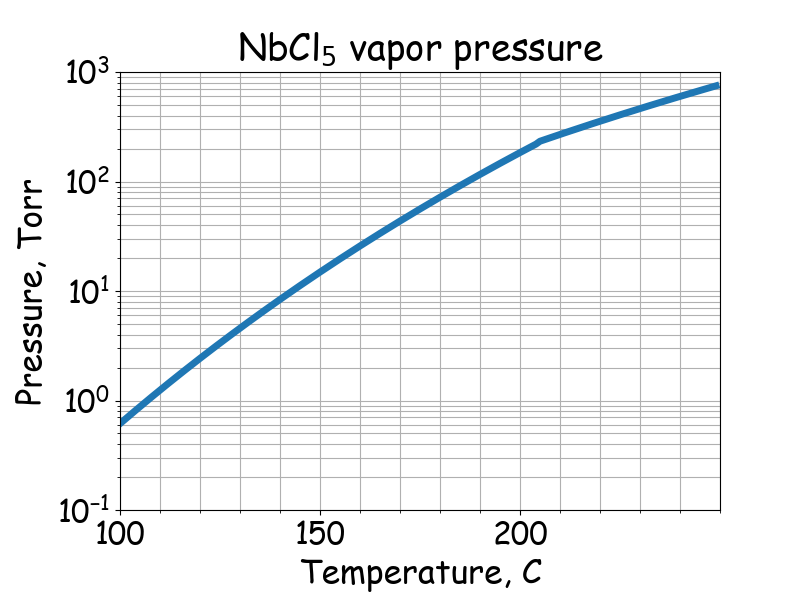
Saturated vapor pressure.
# T -- temperature, C
# P -- saturated vapor pressure, Torr
# Fairbrother, The Chemistry of Niobium and Tantalum, 1967
if 100 <= T <= 205:
P = 10 ** (11.5-4370 / (T + 273.15))
if 205 <= T <= 254:
P = 10 ** (8.37-2870 / (T + 273.15))
Niobium Bromide NbBr5
-
Melting point 267°C
-
Boiling point 361.6°C
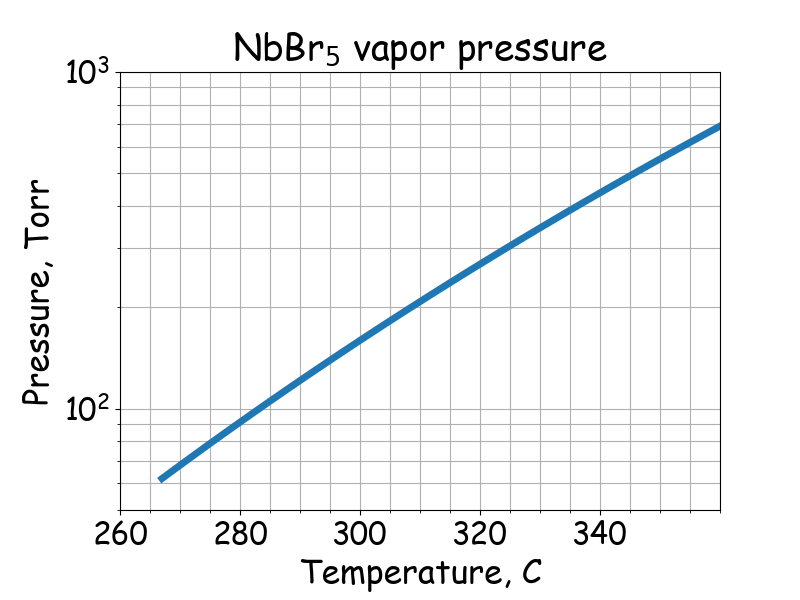
Saturated vapor pressure.
# T -- temperature, C
# P -- saturated vapor pressure, Torr
# Fairbrother, The Chemistry of Niobium and Tantalum, 1967
if 267 <= T <= 361:
P = 10 ** (8.92 - 3850 / (T + 273.15))
Bibliography
-
Fairbrother, F., & Frith, W. C. (1951). 675. The halides of niobium (columbium) and tantalum. Part III. The vapour pressures of niobium (columbium) and tantalum pentafluorides. Journal of the Chemical Society (Resumed), 3051-3056.
-
Junkins, J. H., Farrar Jr, R. L., Barber, E. J., & Bernhardt, H. A. (1952). Preparation and Physical Properties of Niobium Pentafluoride1. Journal of the American Chemical Society, 74(14), 3464-3466.
-
Fairbrother, F., Grundy, K. H., & Thompson, A. (1965). 121. The halides of niobium and tantalum. Part VIII. The densities, viscosities, and self-ionisation of niobium and tantalum pentafluorides. Journal of the Chemical Society (Resumed), 761-765.
-
Alexander, K. M., & Fairbrother, F. (1949). S 48. The halides of columbium (niobium) and tantalum. Part I. The vapour pressures of columbium (niobium) and tantalum pentachlorides and pentabromides. Journal of the Chemical Society (Resumed), S223-S227.
-
Fairbrother, The Chemistry of Niobium and Tantalum, 1967